



The Strategic Use of Ruminally Protected Amino Acids in Dairy Nutrition
The rumen is arguably the most important organ in the ruminant digestive system, writes Adegbola T. Adesogan, University of Florida as part of the proceedings from the University of Florida Dairy Extension 20th Symposium.Introduction
Ruminal microbial protein and volatile fatty acid (VFA) synthesis supply most of the protein and energy needs of the ruminant. Rumen function problems can reduce intake, digestion, and health of ruminants and culminate in death. Furthermore, inefficient rumen function reduces animal performance and health and contributes to environmental pollution.
Feed additives are typically non-nutritive compounds or additives added to diets to improve dietary nutrient utilization, enhance performance, minimize the risk of metabolic diseases, and curtail adverse impacts of diets on the environment.
Specifically, ideal feed additives should have the following attributes:
- Modulate ruminal pH and reduce lactate accumulation.
- Reduce the risk of development of metabolic diseases like diarrhea in neonates and ruminal acidosis or bloat in older livestock.
- Enhance rumen development in neonatal ruminants.
- Improve the efficiency of ruminal energy utilization by reducing ruminal methanogenesis and decreasing the acetate to propionate ratio without reducing milk fat synthesis.
- Improve the efficiency of ruminal nitrogen utilization by (i) reducing proteolysis, peptidolysis, and amino acid deamination, thus minimizing production and losses of NH3 to the environment; (ii) inhibiting the activity of ruminal protozoa that phagocytize desirable bacteria, contribute to proteolysis and deamination, and serve as hosts for methanogens; (iii) enhancing the synthesis of microbial protein by facilitating coupling (synchrony) of ruminal energy and protein supply or by other means.
- Increase ruminal organic matter & fiber digestibility.
- Increase the level and efficiency of animal performance.
- Be cost effective and approved by legislative authorities.
The objective of this paper is to describe dietary additives that exert their main effect on the rumen, to discuss their modes of action, and to summarize the extent to which they meet the criteria listed above. More detailed discussion of the merits of these additives is available in excellent general reviews (Wallace and Newbold, 1995; Hobson and Stewart, 1997; Kung, 2001; Newbold, 2007) as well as reviews on specific additives (ionophores - McGuffey et al., 2001; Duffield et al., 2007a, b; yeasts - Fonty and Chaucheyras-Durand, 2006; Robinson and Erasmus, 2008; Chaucheyras-Durand et al., 2008; enzymes – Beauchemin et al., 2003; Colombatto and Adesogan, 2007; essential oils - Calsamiglia et al., 2007; Benchaar et al., 2008 ).
Ionophores
Ionophores are organic compounds mainly from Streptomyces spp. that facilitate selective transportation of ions across the outer cell membrane. Examples of commonly used ionophores include Rumensin™ (Monensin Sodium), Bovatec™ (Lasalocid Sodium), Salinomycin, and Cattlyst™ (Laidlomycin Propionate Potassium). Although these compounds can no longer be included in animal feeds in Europe, they are approved for preventing coccidiosis, and or improving feed efficiency or performance of different classes of livestock in the US. Monensin, the most widely used of the ionophores, is a polyether antibiotic, which is orally fed as a sodium salt (Hobson and Stewart, 1997; Yang et al., 2007).
Mode of action:
The primary way in which ionophores modify rumen function is by decreasing the ruminal population of gram-positive bacteria relative to that of gram-negative bacteria. Gram-positive bacteria lack the complex cell wall of gram-negative bacteria and the associated lipopolysaccharide layer with its protein channels (porins) that have a size exclusion limit (600 Da) that is impervious to ionophores (> 600 Da; McGuffey et al., 2001). Consequently, ionophores successfully infiltrate the outer cell membrane of gram-positive bacteria and rapidly and repeatedly cause efflux of intracellular K+ from the cell and influx of extracellular protons (Na+ and H+). To overcome the resulting acidity, and the depletion of K+, which inhibits protein synthesis, ATPase pumps are elicited to eject the protons but this depletes energy reserves for bacterial growth. The cytoplasmic acidity culminates in cell death (Guffanti et al., 1979; McGuffey et al., 2001).
Effects on ruminal N and energy utilization
Table 1. Sensitivity of ruminal bacteria to ionophores (Adapted from Hobson and Stewart, 1997).
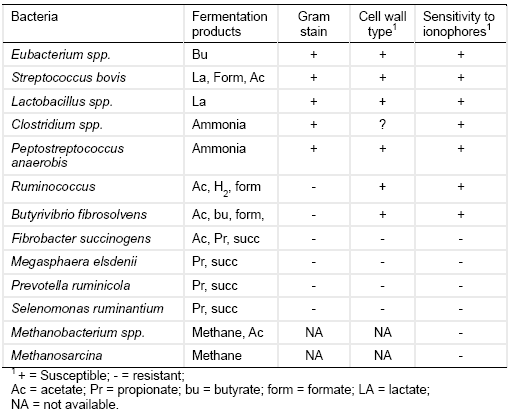
Because gram-positive bacteria mainly ferment dietary nutrients into ‘less desired’ products like acetate, H+, CH4, and NH3 (Table 1), ionophore treatment improves rumen function and animal performance by reducing the production of these metabolites. Ionophore treatment typically reduces the acetate to propionate ratio in the rumen (Table 2) and hence improves the efficiency of ruminal energy utilization. In addition, ionophores like monensin can reduce ruminal methane production by up to 30% (Russell and Strobel, 1989) by inhibiting bacteria providing precursors of methane (formate and H2) rather than directly inhibiting methanogens (Dellinger and Ferry, 1984). For instance methanogenesis by Methanobacterium formicicum was inhibited when the bacteria were grown in a culture enriched with formate but it was largely unaffected when grown in a H2 and CO2 enriched culture (Dellinger and Ferry, 1984). Hydrogen-producing fungi and protozoa may also be inhibited.
Table 2. Effects of monensin treatment on the composition of ruminal fermentation products (Dinius et al., 1976).
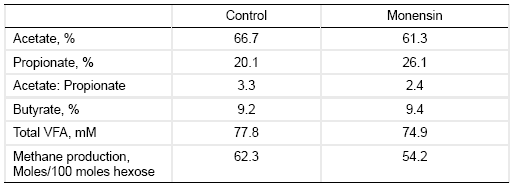
Ionophore treatment often reduces proteolysis slightly, but reduces peptidolysis and amino acid deamination markedly (Hobson and Stewart, 1997). This is often mediated by inhibition of gram-positive hyper ammonia producers such as Peptostreptococcus anaerobius, Clostridium sticklandii, and C. aminopholum. Consequently, ionophore treatment often increases postruminal supply of proteins and peptides.
Effects on animal performance and health
Figure 1. Percentage changes in various responses due to feeding monensin to dairy cows across 77 trials (Duffield et al., 2008)
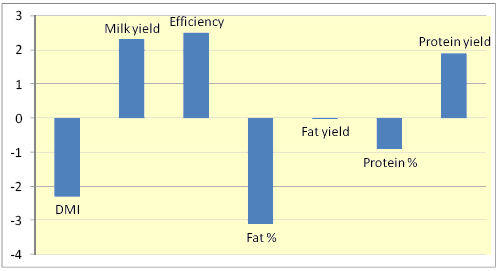
Addition of ionophores to livestock diets has resulted in both negative and positive effects on digestion due to differences in inclusion rate, diet composition, and level of feed intake (McGuffey et al., 2001). Nevertheless, a meta analysis involving 77 dairy cow studies revealed that dry matter intake (DMI), milk fat and protein contents were reduced by 2, 3, and 1%, respectively, whereas milk yield, feed efficiency, and protein yield were each increased by about 2% (Fig. 1).
Ionophores like monensin are very effective at preventing coccidiosis in livestock. By inhibiting the growth of lactate-producing bacteria like Streptococcus bovis and Lactobacillus spp, ionophores can also reduce the risk of ruminal acidosis and bloat. Furthermore, the increased glucose supply resulting from the stimulation of ruminal propionate production by ionophores can reduce the risk of ketosis and fatty liver syndrome in lactating dairy cows (Duffield et al., 2007a).
In summary, ionophores reduce the gram-positive bacteria population in the rumen and this results in greater energetic efficiency (less CH4 & lower acetate to propionate ratio), better protein utilization (less peptidolysis and amino acid deamination, more bypass); less environmental pollution (less NH3 & CH4 production), reduced incidence of coccidiosis, acidosis, bloat, and ketosis; variable effects on fiber digestion, increased efficiency of milk production and decreased milk fat concentration. Monensin has a benefit to cost ratio of 5 to 1 when added to dairy cow diets; it is recommended for increasing feed efficiency in lactating cows and reducing metabolic disorders in dry cows (Hutjens, 2008).
Yeasts
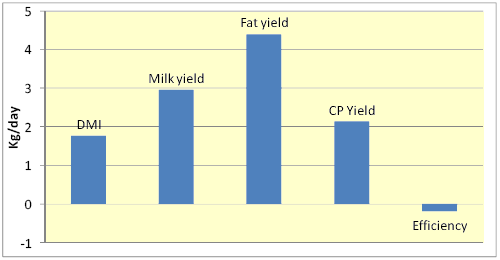
Figure 2. Percentage changes in various responses due to feeding yeasts to dairy cows in 22 trials (Robinson and Erasmus, 2008)
Yeasts are single-celled carbohydrate-fermenting fungi that reproduce by budding. Most commercial products contain a mixture of varying proportions of live and dead Saccharomyces cerevisae cells. Those with a predominance of live cells are sold as live yeasts while others containing more dead cells and the growth medium are sold as yeast cultures (Newbold and Rode, 2006). Examples include Yea-sacc (Alltech Inc.); Levucell SC-20 (Lallemand Animal Nutrition), and Diamond V Yeast culture (Diamond V, Mills Inc.). Yeast cultures have generally regarded as safe (GRAS) status by the US Food and Drug Administration and some have been approved in Europe. The acronym YC will be used subsequently to refer to yeasts and yeast cultures.
Modes of action:
Microbial stimulation: Wallace and Newbold (1993) reported a 50% increase in viable ruminal bacteria in animals fed S. cerevisae across 14 studies. Stimulation of the ruminal fungus Neocallimastix frontalis has also been reported and is partly attributable to provision of thiamine for zoosporogenesis (Chaucheyras-Durand et al., 2005). Furthermore, YC addition has stimulated fiber-digesting bacteria like Fibrobacter succinogens; Ruminococcus spp., and Butyrivibrio fibrosolvens (Weidemeir et al., 1987; and enhanced cell wall colonization by fungi like Neocallimastix frontalis (Chaucheyras- Durand et al., 1995). These factors typically culminate in increases in fiber digestion (Weidemeir et al., 1987; Chaucheyras-Durand et al., 2008), and attendant increases in feed intake.
Oxygen sequestration: Up to 16 L of O2 can enter the rumen daily through water intake, rumination, and salivation (Newbold, 1995) and inhibit the growth of obligate cellulolytic anaerobes like Fibrobacter succinogens (Marounek and Wallace, 1984). Yeasts can make the rumen environment more conducive for anaerobic, autochthonous microbes by scavenging O2 (Chaucheyras-Durand et al., 2008). Many studies have shown that addition of YC decreased the redox potential of the rumen under in vitro and in vivo conditions (Joanny et al., 1998; Chaucheyras-Durand and Fonty, 2002).
pH modulation: Yeasts modulate rumen pH by stimulating entodiniomorphid protozoa that a) engulf starch particles thereby preventing their fermentation to lactate; compete with amylolytic bacteria for starch (Mendoza et al., 1993; Williams and Coleman, 1997), and b) ferment to starch (at a slower rate) to VFA with lower dissociation and acidogenic potential than lactate (Chaucheyras-Durand et al. 2008). These factors collectively reduce the risk of acidosis when YC is supplemented.
Effects on ruminal N and energy utilization
Stimulation of bacterial numbers by YC can increase the rate of substrate fermentation (Ryan and Gray, 1989; Wallace and Newbold, 2006) and microbial protein synthesis. In theory, these factors should increase coupled fermentations that enhance NH3 uptake (Wallace and Newbold, 1995) but results have been inconsistent in practice. Reductions in ruminal NH3 concentration or proteolytic bacteria population have been reported in some studies (Kumar et al., 1994; Chaucheyras-Durand et al., 2005) but not others (Erasmus et al., 1992; Putnam et al., 1997). Wallace and Newbold (1995) noted that fungal additives tend to have small often-insignificant effects on VFA and ruminal ammonia concentrations, and where significant effects occur, they have little biological significance.
Addition of YC to diets or in vitro substrates has decreased methane production in some studies (Lynch and Martin, 2002; Mutsvangwa et al., 1992) but not others (Matthieu et al., 1996; McGinn et al., 2004). These discrepancies may reflect strainspecific effects, dependence on the stage of lactation in trials involving dairy cows, or the inadequate duration (< 48 h) of in vitro studies, which prevent YC from exerting their stimulatory effects on other microbes (Wallace and Newbold, 1995; Newbold & Rode, 2006; Chaucheyras-Durand et al., 2008).
Effects on animal performance.
Addition of YC can increase animal performance but the results seem to vary with the level of performance or stage of lactation (Harris and Lobo, 1988), diet composition (Williams et al., 1991), and management (Wallace and Newbold, 1995). Nevertheless, Wallace and Newbold (1995) reported that across 9 trials, YC treatment produced an average 5.1% increase in milk yield (range = 96 - 117%). Fig. 2 shows that across 22 experiments, addition of yeasts to diets increased DMI and yield of milk and milk components by about 2 to 5% and slightly decreased feed efficiency (Robinson and Erasmus, 2008).
In summary, YC stimulates numbers and activity of total and cellulolytic bacteria, leading to increases in fiber digestion, feed intake, microbial protein synthesis, and animal performance. They also sequester ruminal oxygen, thus facilitating the growth of obligate anaerobes. By stimulating starch-engulfing bacteria that ferment starch to less acidogenic VFA, they can modulate rumen pH and reduce risks of acidosis and bloat. Adding YC has a benefit to cost ratio of 4 to 1 in dairy cow diets; they should be added to diets from 2 weeks prepartum to 10 weeks postpartum or during stressful or off-feed conditions (Hutjens, 2008).
Aspergilius Oryzae
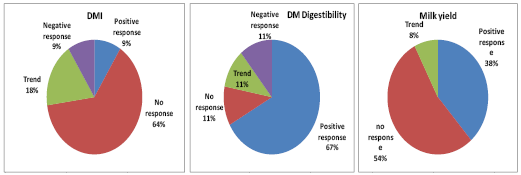
Figure 3. Percentage of successful responses from addition of Aspergillus oryzae to diets across 14 trials
(Queiroz and Adesoogan, 2008, unpublished).
Aspergillus oryzae (AO) fermentation extract consists of fungal spores and mycelium dried onto a wheat bran base (Wallace and Newbold, 1995). The main AO products marketed commercially as feed additives are Amaferm and Vitaferm (Biozyme Enterprises Inc.).
Modes of action:
The exact mode of action of AO is not clear but the following have been proposed:
- Cellulase, xylanase, and esterase enzymes in the extract probably account for the fiber hydrolysis often reported when AO is added to substrates in vitro or to diets (Varel et al.,1993). However, the improved fibrolysis can be diet or forage species specific (Gomez-Alarcon et al., 1990; Beharka and Nagaraja., 1993; Wallace and Newbold, 1995).
- Like yeasts, treatment with AO increases total and cellulolytic ruminal bacteria populations (Wiedmeier et al., 1987; Fondevila et al., 1990) and this has been partly attributed to the dicarboxylic acids in AO extracts (Hobson and Stewart, 1997). Increases in numbers of cellulolytic bacteria also contribute to improved fiber digestion. Effects of AO on anaerobic fungi and protozoa have been inconsistent (Wallace and Newbold, 1995).
- Like yeast extracts, AO extracts are used in the food industry as flavor enhancers, therefore they may increase diet palatability (Wallace and Newbold, 1995).
Effects on ruminal N and energy utilization
Hobson and Stewart (1997) reported that research on microbial feed additives is often frustrating because responses are small and highly variable. Addition of AO has resulted in variable effects on ruminal pH (Weidemeier et al., 1987; Gomez-Alacon et al., 1990), methane production (Martin and Nisbet, 1990; Frumholtz et al., 1989), microbial yield (Wanderley et al., 1987; Gomez-Alacon et al., 1990), amino acid deamination (Frumoltz et al., 1989; Newbold et al., 1993a), and microbial N flow (Gomez-Alacon et al., 1990; Firkins et al., 1990).
Effects on animal performance
In a review of 14 AO publications, we discovered that significant (P<0.05) increases in DMI, DM digestibility and milk yield occurred in 9, 67, and 38% of trials respectively and tendencies (P> 0.5 < 0.1) occurred in 18, 11, and 8%, respectively (Fig., 3). Therefore, AO treatment increased digestibility approximately two thirds of the time and improved milk yield about half of the time (Queiroz and Adesogan, 2008, unpublished). Wallace and Newbold (1995) also reported that across 8 trials, AO treatment produced an average 4.3% increase in milk yield (range = 91 - 110%). Like yeasts, effects of AO also tend to be diet composition and lactation stage dependent (Huber, 1985).
In summary, AO contains fibrolytic enzyme activities that typically improve digestibility and sometimes improve DMI and milk production. In addition, AO can stabilize rumen pH thus reduce the risk of acidosis, bloat, laminitis etc. The benefit to cost ratio of AO is 6 to 1 when fed to dairy cows and it is likely to be most beneficial when acidic diets are fed or under stressful conditions (Hutjens, 2008).
Fibrolytic Enzymes
Enzymes are globular proteins and biological catalysts that increase the rate of reactions or the proximity of reactants. They have been used to increase nutrient availability (e.g. phytase, amylase, and protease) and decrease antinutrients (β- glucanse) in monogastric diets for decades. In contrast, enzyme treatment of ruminant feeds is a relatively novel concept because of their high inherent gastrointestinal hydrolytic capacity (Beauchemin et al., 2003). Various commercial mixtures of cellulase and hemicellulase enzymes with varying endoglucanase, exoglucanase, xylanase, protease, β-glucosidase activities have shown promise at hydrolyzing plant cell walls.
Mode of action:
Fibrolytic enzymes exert their activity through a combination of several pre or post ingestive processes (McAllister et al., 2001). In the pre-ingestive stage, enzyme action is largely effected by direct hydrolysis, which entails cleavage of the glycosidic linkages of the cell wall polysaccharides such that their constituent monomeric units are released. The process removes structural barriers restricting accessibility to the more digestible cell contents within the forages. In the post-consumptive stage, enzyme action is thought to be mediated through direct hydrolysis, improved palatability probably due to the increased production of mono and disaccharides, reduced viscosity (Rode et al., 1999), increased proximity between substrates and microbes, synergy between ruminal fibrolytic and exogenous enzymes (Morgavi et al. 2000), and stimulation of bacterial activity and attachment by exogenous enzymes and other substances introduced with the enzyme complex (Beauchemin et al., 2003).
Effects on ruminal energy and N utilization
Most studies involving enzyme treatment of feeds report increases in either the rate (Beauchemin et al., 2003) or extent (Krueger et al., 2008) of fiber digestion, or a reduction in fiber concentration (Krueger and Adesogan, 2008). These effects have sometimes decreased the acetate to propionate ratio (Dean et al., 2003; Krueger and Adesogan, 2008) but reductions in deamination and methane production or increases in proteolysis, and microbial protein yield or flow are not consistent.
Effects on animal performance
Table 3. Effect of enzyme treatment of feeds on milk production in studies published between 1999 and 2000 (Kung, 2001).

Several studies have shown improvements in DM or fiber digestibility and or reductions in cell wall content following enzyme treatment of forages at the point of conservation (Adogla Bessa et al., 1999, Schingoethe, 1999) and just prior to feeding (Rode et al., 1999; Beauchemin et al., 1999; Kung et al., 2000; Yang et al., 2000) but others have not (Hristov et al., 1998; Lewis et al., 1999; Knowlton et al., 2002). Dry matter intake in dairy cows has been increased (Rode et al., 1999; Schingoethe et al., 1999; Yang et al., 1999; 2000; Beauchemin et al., 2000; Vicini et al., 2003) or unaffected (Beauchemin et al., 1999; Lewis et al., 1999; Kung et al., 2000) and changes milk yield and composition have been inconsistent (Beauchemin et al., 1999; Lewis et al., 1999; Schingoethe et al., 1999; Rode et al., 1999; Yang et al., 1999; 2000; Kung et al., 2000; Vicini et al., 2003). Nevertheless, (Newbold, 1997) reported that fibrolytic enzyme treatment improves average daily gain (ADG) in beef cattle by 5 to 30% and increases milk yield by 2 to 15%. Table 3 also itemizes increases in milk production caused by enzyme application to feeds; on average, milk production was increased by 1.3 kg/d by fibrolytic enzyme addition to diets (Kung, 2001). However, a recent review revealed that many of those improvements were not statistically significant (P<0.05). Based on a review of 41 treatments from 19 published experiments conducted between 1999 and 2006, supplementation with fiber-digesting enzymes increased milk production by 2.3-2.7 kg/day when effective (P < 1.0), but this only occurred in 40% of the trials examined (Adesogan et al., 2007). Nevertheless, some of the numerical (P>0.01) responses may have masked real effects because lack of significance in additive evaluation trials sometimes reflects inadequate replication of experimental units and the consequent lack of power to detect treatment differences.
Discrepancies among enzyme evaluation trials are attributable to animal factors such as the performance level as well as differences in enzyme activity, substrate type, prevailing temperature and pH, treatment duration, the method and medium of application (Colombatto et al., 2003), the presence of co-factors or inhibitors and the enzyme to substrate ratio. Recent attempts at improving the potency of fibrolytic enzymes have focused on two main approaches:
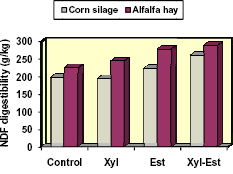
Figure 4. Synergistic effects of applying a xylanase (xyl) and esterase (est) enzyme on the NDF digestibility (g/kg) of corn and alfalfa hay (Eun and Beauchemin, 2006)
-
Using rumen-like conditions: Colombatto et al. (2003) emphasized the importance of testing and determining fibrolytic activities of enzymes under rumenlike pH and temperature because activities reported on enzyme labels are often done under different conditions. The optimal pH for most enzymes is 4 – 5.5, which is notably lower than the typical rumen pH. Vicini et al. (2003) conducted what was probably the largest scale fibrolytic enzyme trial and concluded that the enzymes they tested were ineffective at improving the performance of dairy cows because their activity was relatively low under rumen pH and temperature. Fibrolytic enzymes selected for high activity under rumen conditions are more likely to be effective at enhancing fiber utilization in ruminants.
-
Strategically combining key fibrolytic enzyme activities: Enzymes are highly specific in both binding chiral substrates and catalyzing reactions due to geometric and stereochemical complementarity between enzymes and substrates (Voet and Voet, 2003). Most of the previous fibrolytic enzyme research in ruminant nutrition involved only cellulase and xylanase enzymes, which do not hydrolyze etherified or esterified ferulic and coumaric acid linkages that bind digestible arabinoxylans to lignin in plant cell walls. Such linkages have been compared to the molecular equivalent of spot-welding on a steel mesh frame (Iiyama et al., 1994). Researchers have recently begun to pay closer attention to targeting such digestion-impeding cell wall components. Ferulic acid esterases (FAEs) can release ferulic acid (FA) bound to arabinose side chains of hemicellulose (Faulds and Williamson, 1994), allowing further degradation of the cell wall by other polysaccharidases. Addition of FAE to other cell wall-degrading enzymes, like xylanase and cellulase, produces a synergistic effect on degradation of plant cell walls (Faulds and Williamson, 1995; Bartolome et al., 1997; Rodrigues et al., 2001; Yu et al., 2002, 2003, 2005; Eun and Beauchemin, 2007; Eun et al., 2007), due to increased accessibility to digestible cell wall components by rumen microorganisms. Our recent results (Krueger et al., 2004; 2008; Adesogan et al., 2005; Krueger and Adesogan, 2008) also confirm these findings.
Eun and Beauchemin (2006) evaluated the efficacy using different mixtures of enzymes having predominantly xylanase or esterase activity to improve the digestion of alfalfa hay and corn silage. They identified a mixture that increased the in vitro digestibility of DM and NDF of the respective forages by 7 – 9% and 28 - 31% (Fig. 4). We examined the performance of dairy cows fed low- (33%) or high- (48%) concentrate, corn silage-based diets with or without this same enzyme mixture (Adesogan et al., 2007). Enzyme addition increased the efficiency of milk production; increased milk production by 2.7 kg/d in cows fed the high concentrate diet, and produced a nonsignificant increase in those fed the low concentrate diet. These studies show potential benefits of strategically mixing different fibrolytic enzyme activities.
Under exploited approaches to improving fibrolytic enzyme activity include the following:
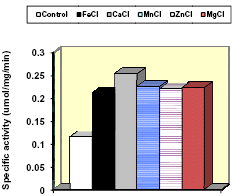
Figure 5. Effects of adding cofactors on esterase activity on a fibrolytic enzyme (Gonzalez and Adesogan, 2008, unpublished)
-
Addition of Cofactors: The activity of many enzymes is dependent on the presence of cofactors or coenzymes. Key cofactors for fibrolytic enzymes were listed by Brenda (2008). Nevertheless, we found no published studies that evaluated effects of adding cofactors to increase hydrolysis of ruminant feeds by fibrolytic enzymes. Our preliminary studies (Gonzalez and Adesogan, 2008, unpublished data) revealed that addition of cofactors doubled the activity (Fig. 5) of the same xylanase-esterase enzyme mixture that had increased the level and efficiency of milk production by dairy cows (Adesogan et. al, 2007). More work is needed on using cofactors to improve fibrolytic enzyme activity.
-
Exploiting novel enzyme activities: In a quest to find enzymes capable of releasing ferulic acid from vegetable cell walls to stimulate insulin production and alleviate symptoms of diabetes, Lai et al. (2008, unpublished), isolated a strain of lactic acid bacteria displaying strong ferulic acid esterase activity from the stool of genetically susceptible but diabetes-resistant rats. These investigators purified and characterized the esterase enzyme, and recently showed that it had far greater esterase activity than the xylanase esterase mixture that improved the level and efficiency of milk production in our experiment (Adesogan et al., 2007). One of the most promising microbes for ethanol production was isolated from a xylivorous beetle (Suh et al., 2003); consequently, there is interest in mining microbes from xylivorous fish for biomass hydrolysis and conversion to ethanol (Bjorndal et al., 2008, personal communication). Enzymes from such microbes are likely to be effective in hydrolyzing plant cell walls.
In summary, fibrolytic enzymes increase fiber digestion in ruminant diets through a combination of pre and post ingestive processes that enhance cell wall hydrolysis. However, they have had inconsistent effects on animal performance. The benefit to cost ratio of such enzymes is 2 to 3:1 (Hutjens, 2008). More research is required to provide consistent, economically attractive improvements in animal performance from fibrolytic enzymes.
Essential oils
Table 4. Sources and constituents of some essential oils (Adapted from Chao et al., 2000 and Benchaar et al., 2008)
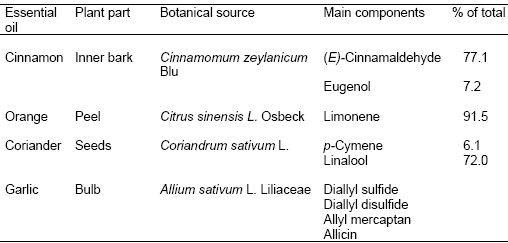
Essential oils (EO) are fragrant, complex, volatile, secondary metabolites extracted by distillation from various parts of plants (Yang et al., 2007). Chemically, they are not true oils, rather, they are variable mixtures consisting principally of terpenoids, mainly monterpenes (C10), sesquiterpenes (C15) and various other compounds (Benchaar et al., 2008). The composition of EO varies with the species, botanical fraction, maturity, and environment of the plant. Several EO have strong antimicrobial properties against various microorganisms, therefore, they have been evaluated as alternatives to antibiotic drugs and additives in human and animal diets. Table 4 lists some common EO and their active ingredients.
Mode of action:
The following theories have been propounded to explain the action of EO:
-
Essential oils modulate cellular targets particularly by interacting with processes associated with the cell membrane such as ion gradients, protein translocation, phosphorylation, ATP production etc (Dorman and Deans, 2000; Acamovic and Brooker, 2005, as cited by Benchaar et al., 2008). The hydrophobic, lipophilic nature of EO contributes to this effect (Benchaar et al., 2008).
-
Some studies have shown that like monensin, EO selectively inhibit gram-positive bacteria (Burt, 2004; Trombetta et al., 2005). However, Benchaar et al. (2008) cited studies suggesting that the small molecular weight of EO also allows them to penetrate the cell membrane of gram-negative bacteria; for instance, thymol and caravacrol have inhibited gram-negative bacteria (Helander et al., 1998).
Effects on ruminal N and energy utilization.
Due to the wide range of EO in nature, their effects on rumen fermentation vary. Some EO have monensin-like effects on gram positive bacterial populations and ruminal VFA proportions, whereas others have more general inhibitory effects on ruminal bacteria such that total VFA production was reduced (Benchaar et al., 2008). These differences partly reflect variations in the chemical structure of the EO. Oxygenated monoterpenes have strongly inhibited bacterial activity, whereas monoterpene hydrocarbons either slightly inhibited or stimulated bacterial activity (Benchaar et al., 2008).
Benchaar et al (2008) cited studies showing that anise, cade, capsicum, cinnamon, clove, dill, garlic, eugenol, and cinnamaldehyde and their active ingredients reduced amino acid deamination considerably. They, attributed this to a monensin-like inhibition of some hyper-ammonia producing bacteria such as Clostridium sticklandii and Peptostreptococcus anaerobius. However, amino acid deamination was unaffected by EO addition in some studies (Castijellos et al., 2007, Benchar et al., 2007). Essential oil addition resulted in deamination when a low protein diet rather than a high protein diet was fed. These factors reflect dependence of the outcome on the nature of the diet (Wallace, 2004), the dose rate (Busquet et al., 2006; Benchaar et al., 2008), and the type of EO. Furthermore, the high dose rates at which, deamination was evident in some studies also inhibited VFA production, which could compromise energy supply to the ruminant. In contrast, some studies have reported that EO addition increased VFA production (Castillejos et al., 2005; Benchar et al., 2007), whereas others showed no effect (Newbold et al., 2004; Beauchemin and McGinn, 2006), further reflecting dependence of the outcome on the dose and diet (Benchaar et al., 2008).
Addition of encapsulated horseradish, garlic oil, and cinnamaldehyde resulted in beneficial reductions in the acetate to propionate ratio of ruminal fluid but also increased butyrate proportion (Mohammed et al., 2004; Busquet et al. (2005; 2006). However, studies with eugenol resulted in lower propionate concentration (Castillejos et al., 2006). One of the challenges with EO evaluation is that microbes can adapt to EO, therefore the short-term in vitro experiments that have been used in several studies on EO are inappropriate evaluation tools (Benchaar et al., 2008).
Some EO, particularly garlic extracts are effective at curtailing methane production in the rumen (Busquet and Martin, 2000; Busquet et al. (2005a). Reductions of up to 70% by garlic oil or diallyl disulfide, one its main components have been reported. These reductions exceeded that achieved with monensin (Busquet et al., 2005b; Benchaar, et al., 2008) and they were attributed to direct inhibition of methanogenic archaea rather than to inhibition of precursors of methane. Other EO like thymol and clove or fennel extracts have also reduced methane production but also reduced digestibility or propionate concentration (Evans and Martin, 2000; Patra et al., 2005).
Effects on animal performance.
Few studies on effects of EO supplementation on animal performance have been published; many showed no effects on milk yield or composition (Yang et al., 2007; 2007), though one showed an improved gain to feed ratio (Benchar et al., 2006) in beef cattle.
In summary, EO vary considerably in chemical structure, source, and activity. Consequently, their effects on ruminal fermentation and animal performance are inconsistent. More research is needed to identify EO that only have desirable effects on rumen function and animal performance.
Other Additives
Buffers
Buffers are weak acids or alkalis that resist changes in H+ concentration or pH. They are added to diets to complement the buffering effect of saliva and neutralize ruminal acidity. Consequently, buffer addition reduces the risk of acidosis in cattle fed starch-rich diets or acidic silages, and decreases the incidence of bloating in cattle fed spring grass/legume pastures. Examples include sodium bicarbonate, limestone, sodium bentonite, and magnesium oxide.
The main mode of action of buffers involves increasing pH or resisting a change in pH. Higher pH values facilitate fiber digestion, hence buffer addition has increased the acetate to propionate ratio in the rumen. In addition, certain buffers increase ruminal osmolality and thereby increase the ruminal fluid outflow rate, which is associated with reduced ruminal propionate proportion and hence, increased milk fat synthesis (Rogers et al., 1982; Hobson and Stewart, 1997).
In summary, buffers stabilize rumen pH thereby prevent acidosis, bloat, rumenitis, and laminitis. They also enhance water intake, ruminal fluid outflow, fiber digestion and milk fat synthesis. Buffers are recommended for stabilizing ruminal conditions when acidic or bloat-inducing diets are fed.
Organic acids
Dicarboxylic organic acids like aspartate, malate, and fumarate have been evaluated as feed additives because of their potential to reduce methanogenesis by ‘sinking’ H2 during their conversion to propionate (Newbold and Rode, 2006). This theory has been validated in several studies (Bayaru et al., 2001; Moss and Newbold, 2002; Wallace et al., 2005). The increased H2 removal could also stimulate cellulolytic bacteria and increase cellulose digestion (Newbold and Wallace, 2006). However, such acids have had inconsistent effects on animal performance (Sanson and Stallcup, 1984; Martin et al., 1999). Although these organic acids have GRAS status, problems with palatability (Moss and Newbold, 2002), decreased ruminal pH (Asanuma et al., 1999), inconsistent responses, and high costs (Newbold and Rode, 2006), have limited their adoption.
Conclusion
Feed additives can be used to manipulate rumen function, increase the level and efficiency of animal performance, and minimize adverse effects of diets on animal health and the environment. However, various products with contrasting effects are available. Hence, careful scrutiny of the literature is required to identify effective additives. Only research-proven additives that consistently produce an economically justifiable return should be added to diets.
Further Reading
| - | You can view a list of the full IFAS 20th Symposium proceedings by clicking here. |
February 2009


