



Use of Ultrasonography to Age Pregnancy in Early Gestation in Dairy Cows
The use of ultrasound to monitor the development of calves in dairy cattle is becoming more and more popular acording to a study by Klibs N.A. Galvão, DVM, MPVM, PhD, assistant professor in the Department of Large Animal Clinical Sciences, College of Veterinary Medicine in the University of Florida.Introduction
Using ultrasonography for diagnosis and aging of pregnancy is becoming more widespread among bovine practitioners (Fricke 2002; Fricke and Lamb 2005), even though many practitioners still commonly use palpation per rectum (Mortimer and Hansen 2006; Ramano et al. 2006). Palpation per rectum is most commonly performed after 35 days post-breeding, when it is most accurate (Kastelic et al. 1988; Mortimer and Hansen 2006; Ramano et al. 2006). One of the main advantages of using ultrasonography is early detection of pregnancy, usually after 27 days post-breeding (Kastelic et al. 1988; Beal, Perry, and Corah 1992; Fricke 2002; Jones and Beal 2003; Fricke and Lamb 2005; Medan and Abd El-Aty 2010; Ramano et al. 2006). Other advantages of ultrasonography include monitoring fetal development and viability, ovarian structures, uterine anatomy, involution, and pathology, as well as determining fetal age and sex (Jones and Beal 2003; Stroud 2006).
Aging pregnancy using fetal measurements has proven to be accurate (Jones and Beal 2003; Mortimer and Hansen 2006; Ribadu and Nakao 1999). Calving was predicted within an average of 4.5 days when crown to rump length was used before 55 days of gestation, and it was predicted within 6.9 or 7.8 days when either the head or the trunk was used after 55 days of gestation (Jones and Beal 2003; Lamb and Fricke 2004). Guidelines to age pregnancy in early gestation exist, but they are not very detailed (Youngquist and Threlfall 2007; Poock and Wilson 2011). In Youngquist and Threlfall (2007), Table 39-1 states that the size of amniotic vesicle (AV) ranges from 0.8 to 1.0 cm at 30 days of gestation and from 2.0 to 3.0 cm at 40 days of gestation. Poock and Wilson (2011) present a table adapted from Drs. Jill Colloton and Brad Stroud where the crown to rump measurement is 0.9 cm before 30 days of gestation, 1.0 cm at 35 days of gestation, and 2.0 cm at 40 days of gestation. This publication presents a detailed measurement of the amniotic vesicle from 27 to 34 days after breeding so a more precise aging of early pregnancies can be performed.
Materials and Methods
The amniotic vesicle was measured at its widest length using the grid available in the ultrasound machine Easi-Scan 3 (BCF Technology Ltd, Livingston, UK) in 167 pregnancies between 27 and 34 days of gestation. Descriptive statistics and a linear regression analysis were performed using the MINITAB® (State College, PA, USA) software.
Results
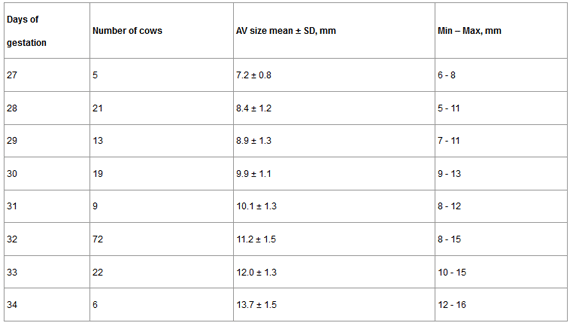
The average size of the AV is presented in Table 1. The size of the AV ranged from 7.2 to 13.7 mm from 27 to 34 days of gestation.
Table 1. Descriptive statistics for amnotic vesicle size from 27 to 34 days of gestation.
The linear regression analysis is shown in Figure 1.
Linear Regression Analysis of Amniotic Vesicle (AV) size by Gestational Age (GA).
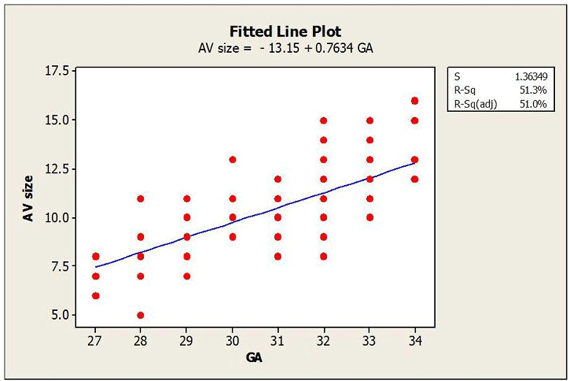
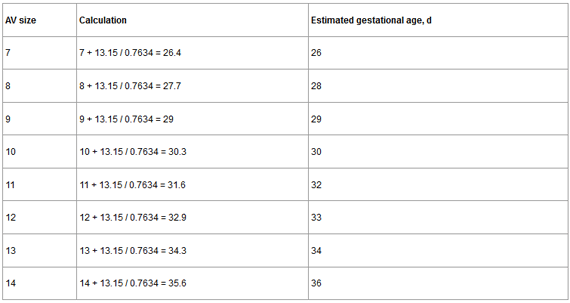
The linear regression equation is AV size = -13.15 + 0.7634 x days of gestation. To estimate the day of gestation, you need to use this equation: days of gestation = AV size + 13.15/0.7634. An example with different AV sizes is shown in Table 2.
Table 2. Estimation of gestation age based on linear regression equation.
Using This Method in the Field
For each additional day of gestation from 27 to 34 days, AV size increases by roughly 1 mm. To make this method easy to implement in the field, a good rule of thumb would be to add 20 mm to the size of the amniotic vesicle. For example, if the AV size is 8 mm, the estimated gestational age would be 28 days. This practical recommendation would be fairly accurate and easy to implement.
References
Beal, W.E., R.C. Perry, and L.R. Corah. 1992. “The Use of Ultrasound in Monitoring Reproductive Physiology of Beef Cattle.” J. Anim. Sci. 70: 924-9.
Fricke, P.M. 2002. “Scanning the Future — Ultrasonography as a Reproductive Management Tool for Dairy Cattle.” J. Dairy Sci. 85:1918-26.
Fricke, P.M., and G.C. Lamb. 2005. “Potential Applications and Pitfalls of Reproductive Ultrasonography in Bovine Practice.” Vet. Clin. North Am. Food Anim. Pract. 21: 419-36.
Jones, A.L., and W.E. Beal. 2003. “Reproductive Applications of Ultrasounds in the Cow.” Bovine Practitioner 37:1.
Kastelic, J.P., S. Curran, R.A. Pierson, and O.J. Ginther. 1988. “Ultrasonic Evaluation of the Bovine Conceptus.” Theriogenology 29:39-54.
Lamb, G.C., and P.M. Fricke. 2004. “Ultrasound – Early Pregnancy Diagnosis and Fetal Sexing." Proceedings, Applied Reproductive Strategies in Beef Cattle, 219-29.
Medan, M.S., and A.M. Abd El-Aty. 2010. “Advances in Ultrasonography and Its Applications in Domestic Ruminants and Other Farm Animals Reproduction.” Journal of Advanced Research 1:123-8.
Mortimer, R., and T. Hansen. 2006. “The Future of Pregnancy Testing in Beef Cattle.” Proceedings, Applied Reproductive Strategies in Beef Cattle, 281-8.
Poock, S.E., and D.J. Wilson. 2011. “A Review of the Use of Ultrasound for Reproductive Purposes in Beef Cattle.” Proceedings, Applied Reproductive Strategies in Beef Cattle, 231-5.
Ramano, J.E., J.A. Thompson, D.W. Forrest, M.E. Westhusin, M.A. Tomaszweski, and D.C. Kraemer. 2006. “Early Pregnancy Diagnosis by Transrectal Ultrasonography in Dairy Cattle.” Theriogenology 66:1034-41.
Ribadu, A.Y., and T. Nakao. 1999. “Bovine Reproductive Ultrasonography: A Review.” Journal of Reproduction and Development 45:13-28.
Stroud, B. 2006. “The Use of Ultrasound in Cow/Calf Applications.” Proceedings, American Association of Bovine Practitioners 39:3-7.
Youngquist, R.S., and W.R. Threlfall. 2007. “Pregnancy Diagnosis.” In Current Therapy in Large Animal Theriogenology, 2nd Edition, edited by R.S. Youngquist and W.R. Threlfall, 298-9. St. Louis, MO: Saunders Elsevier.
June 2012


